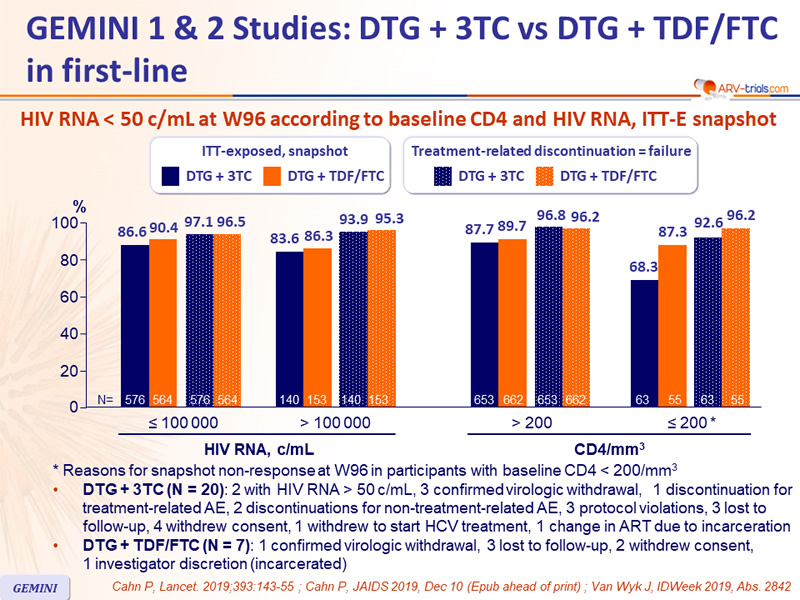
#Gemini 2 trial trial#
“Our wholly-owned pipeline continues to make progress with the recent initiation of our imsidolimab GEMINI-1 Phase 3 trial in GPP and anticipated initiation of our rosnilimab AZURE alopecia areata Phase 2 trial during the remainder of 2021,” said Hamza Suria, president and chief executive officer of AnaptysBio. (Nasdaq: ANAB), a clinical-stage biotechnology company developing first-in-class antibody product candidates focused on emerging immune control mechanisms applicable to inflammation and immuno-oncology indications, today reported operating results for the third quarter ended Septemand provided pipeline updates.
04, 2021 (GLOBE NEWSWIRE) - AnaptysBio, Inc. JEMPERLI royalty monetization transaction signed with Sagard Healthcare Royalty Partners for $250 million upfront payment in exchange for capped return on royalties and certain milestones below $1 billion in annual salesĮxpect to end 2021 with approximately $600 million in cash and will continue to operate in a capital-efficient manner JEMPERLI (dostarlimab) was approved for mismatch repair deficient (dMMR) recurrent or advanced solid tumors in the US, earning $20 million cash milestone during Q3 2021 Rosnilimab Phase 1 healthy volunteer top-line data and initiation of AZURE Phase 2 trial in alopecia areata anticipated in Q4 2021

Top-line data from ongoing imsidolimab ACORN Phase 2 trial in moderate-to-severe acne anticipated in H1 2022 and HARP Phase 2 trial in moderate-to-severe hidradenitis suppurativa in H2 2022 Imsidolimab GPP GALLOP Phase 2 16-week data presented at 2021 EADV Congress and GEMINI-1 Phase 3 trial initiated subsequent to FDA end-of-Phase 2 meeting
